Following the news from Europe
Provided by AGPBiomed Industries, Inc. Advances Dual-Track Obesity Therapy Strategy with Near-Term Revenue and NA-931 Phase 3 Launch
Biomed Industries, Inc. Advances Dual-Track Obesity Therapy Strategy with Near-Term Revenue from Bioxentra Life Sciences, Inc. and NA-931 Phase 3 Launch
Winning in this market requires both speed and differentiation. Our model generates near-term revenue while advancing a potentially best-in-class therapy to drive scalable, long-term value creation.”
SAN JOSE, CA, UNITED STATES, May 5, 2026 /EINPresswire.com/ -- — Biomed Industries, Inc. today announced that CEO Dr. Lloyd L. Tran presented new clinical data on NA-931 at the Metabolic Medicine Summit (April 29–30, 2026, Boston), alongside a strategic licensing agreement with Bioxentra Life Sciences, Inc.— Dr. Lloyd L. Tran, CEO of Biomed
Summary of Phase 2 Clinical Results:
NA-931, a novel oral quadruple receptor agonist, demonstrated strong results in a 13-week Phase 2 study (n=125), achieving up to 13.8% mean weight loss, compared to approximately 2% with placebo, with 72% of patients reaching ≥11% weight loss.
The therapy was generally well tolerated. Most gastrointestinal (GI) adverse events were reported as insignificant or mild, with 83% classified as insignificant. Mild nausea and vomiting occurred in 7.3% of treated subjects, and diarrhea in 6.3%. No muscle loss was observed, and no clinically meaningful differences in GI-related adverse events were reported versus placebo. Unlike many existing therapies, NA-931 is designed to promote weight loss while preserving muscle mass and improving tolerability.
A Time-Sensitive Market Opportunity:
The obesity therapeutics market is undergoing rapid structural change. Pricing pressure on oral GLP-1 therapies and the anticipated loss of patent protection for semaglutide in 2026 in several countries are expected to accelerate competition, expand access, and compress margins—creating a limited window for new entrants to establish market position.
Dual-Track Strategy: Near-Term Revenue and Long-Term Upside:
Biomed Industries and Bioxentra are deploying a capital-efficient, dual-track model designed to capture value across both timelines:
• Near-term monetization: Launch of Bioxentra as a nutraceutical platform, enabling rapid market entry and early revenue generation
• Pipeline advancement: Continued development of NA-931 toward Phase 3, targeting a differentiated profile with muscle preservation and improved tolerability
• Capital efficiency: Early commercial cash flow supporting ongoing clinical development
“Winning in this market requires both speed and differentiation." said Dr. Tran. "Our business model generates near-term revenue while advancing a potentially best-in-class therapy to drive scalable, long-term value creation.”
Positioned Alongside GLP-1 Therapies:
GLP-1 therapies such as semaglutide and tirzepatide have validated the market but leave important gaps, including muscle loss, tolerability challenges, and post-treatment weight regain.
Real-world data indicate that approximately 50% or more of patients discontinue GLP-1 therapies within the first year, with some studies reporting discontinuation rates approaching 70% or higher over longer follow-up periods, driven in part by adverse events, cost, and access limitations.
NA-931 is a first-in-class oral quadruple receptor agonist targeting insulin-like growth factor-1 (IGF-1), glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors. This multi-target mechanism is designed to enhance metabolic regulation while supporting muscle preservation.
Bioxentra is designed as a metabolic companion platform that can be used:
• Alongside GLP-1 therapies to support muscle preservation during weight loss
• Prior to initiation to optimize metabolic readiness
• Following discontinuation to support weight maintenance and reduce rebound
This positioning enables participation across the full treatment lifecycle, addressing real-world adherence challenges and expanding the total addressable market beyond single-drug use.
Investor Value Proposition:
This integrated strategy aligns with key investor priorities:
• Faster time-to-revenue compared to traditional drug-only models
• Reduced clinical risk exposure through parallel commercialization
• Large and expanding total addressable market (TAM) driven by global obesity demand
• Multiple value inflection points, including nutraceutical launch, Phase 3 advancement, and potential regulatory milestones
By combining near-term commercial traction with a high-value late-stage asset, Biomed Industries and Bioxentra are positioned to capture both short-term revenue growth and long-term pharmaceutical value in a rapidly expanding healthcare sector.
About Biomed Industries, Inc.
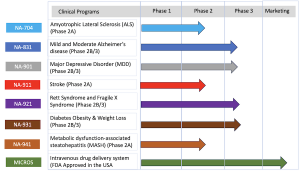
Biomed Industries, Inc. is a pioneering biopharmaceutical company committed to developing novel therapeutics that address unmet medical needs. Its innovative research platform has produced treatments for conditions including Alzheimer’s disease, ALS, Traumatic Brain Injury, Major Depressive Disorder, Diabetes, Obesity, MASH, Stroke, and rare diseases like Rett Syndrome. (www.biomedind.com )
About Bioxentra Life Science, Inc.
Bioxentra™ is a life sciences company dedicated to advancing human health through the discovery and development of science-based nutraceuticals and bioactive compounds. Bioxentra's science-based metabolic companion platform is designed to enhance GLP-1 therapy, improve tolerability, and support long-term weight maintenance. (www.bioxentra.com )
Media Contact:
For more information, please contact:
Michael Willis
Vice President
Biomed Industries, Inc.
Tel. 800-824-5135
Direct: 303-717-0464
MWillis@biomedind.com
www.biomedind.com
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of applicable securities laws. These statements include, but are not limited to, statements regarding the development, clinical progress, regulatory pathway, commercialization potential, and market opportunity of NA-931 and Bioxentra’s metabolic platform, as well as anticipated market dynamics in the obesity therapeutics sector.
Forward-looking statements are based on current expectations, assumptions, and beliefs and are subject to significant risks and uncertainties that could cause actual results to differ materially. These risks include, but are not limited to, the outcome of ongoing and future clinical trials, regulatory approvals, market adoption, competitive developments, pricing pressures, and general economic conditions.
Biomed Industries, Inc. undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise, except as required by law.
Michael Willis
Biomed Industries, Inc.
+1 800-824-5135
email us here
Visit us on social media:
LinkedIn
X
Biomed introduction video
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.